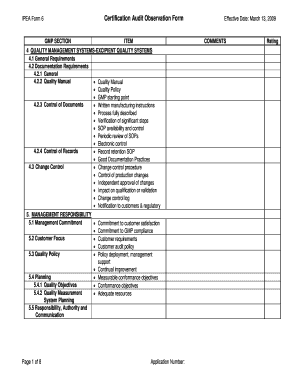
Get Certification Audit Observation Form
How It Works
-
Open form follow the instructions
-
Easily sign the form with your finger
-
Send filled & signed form or save
Experience a faster way to fill out and sign forms on the web. Access the most extensive library of templates available.
Compendia FAQ
Use professional pre-built templates to fill in and sign documents online faster. Get access to thousands of forms.
Keywords relevant to Certification Audit Observation Form
- nonconforming
- Recirculation
- reprocessed
- compendial
- reusable
- unlabeled
- Compendia
- bse
- SOPs
- crosscontamination
- conformance
- reprocessing
- retrievable
- inputs
- numbering
USLegal fulfills industry-leading security and compliance standards.
-

VeriSign secured
#1 Internet-trusted security seal. Ensures that a website is free of malware attacks.
-
Accredited Business
Guarantees that a business meets BBB accreditation standards in the US and Canada.
-

TopTen Reviews
Highest customer reviews on one of the most highly-trusted product review platforms.



